London Dispersion Forces Co2

At room temperature neopentane c 5 h 12 is a gas whereas n pentane c 5 h 12 is a liquid.
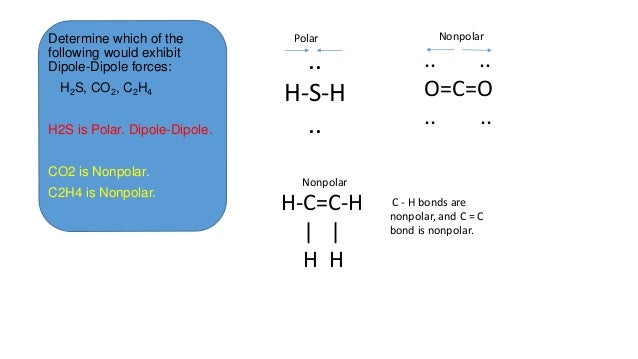
London dispersion forces co2. Hydrogen bonding london dispersion forces elemental iodine i2 is a solid at room temperature. Yes ch4 only has london dispersion forces present because ch4 is a non polar molecule and non polar molecule has london dispersion forces present in them. London dispersion forces are part of the van der waals forces or weak intermolecular attractions. Asked in chemistry chemical bonding.
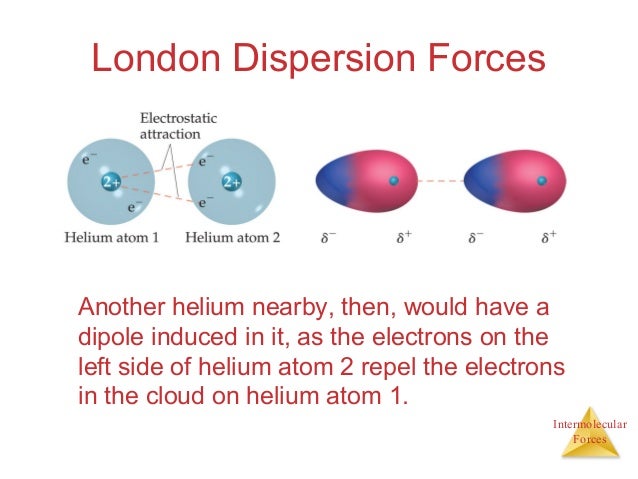
London dispersion forces tend to be. They will all have london dispersion forces the ones that have dipole dipole forces with the molecules that actually have dipole moments and a overall net charge. Molecular shape the shapes of molecules also affect the magnitudes of dispersion forces between them. Dipole dipole ion dipole london dispersion interactions duration.
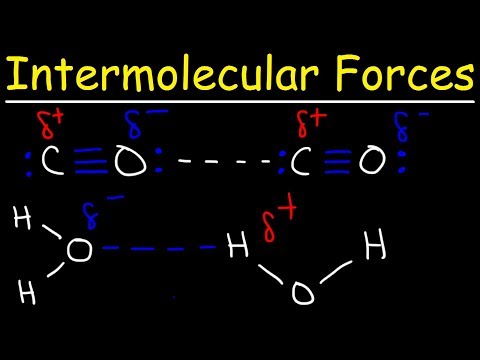
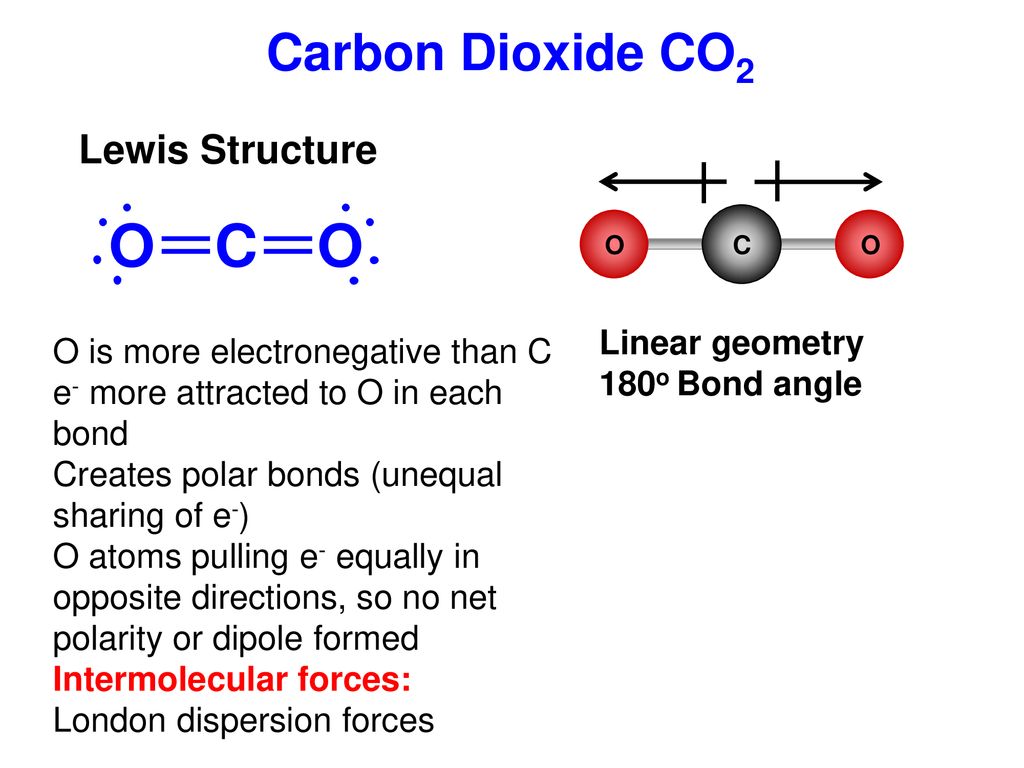
Co2 is a nonpolar molecule thus having dispersion london forces. Question 9 which one of the following interactions are present between co2 molecules. This video discusses the intermolecular forces of carbon dioxide co2. Stronger between molecules that are easily polarized.
I presume you mean co2 with co2 and br2 with br2 and not mixtures of co2 and br2 for example. These london dispersion forces are often found in the halogens e g f 2 and i 2 the noble gases e g ne and ar and in other non polar molecules such as carbon dioxide and methane.